
TIRZEPAT
10 mg | 20 mg
Metabolic & Weight Management
TIRZEPAT is a modern pharmaceutical-grade injectable peptide developed in the United States for medical and professional use. It is intended for the management of carbohydrate metabolism disorders and body weight control in adults.
When TIRZEPAT is selected:
In type 2 diabetes mellitus to improve glycemic control
In overweight or obesity
In metabolic syndrome
To reduce appetite and support correction of eating behavior
When long-term stabilization of blood glucose levels is required
Mechanism of action:
TIRZEPAT simultaneously activates both GIP and GLP-1 receptors, resulting in:
Glucose-dependent enhancement of insulin secretion
Reduction of glucagon secretion
Delayed gastric emptying
Decreased hunger sensation and increased satiety
Due to albumin binding, the elimination half-life is approximately 5 days, allowing for once-weekly administration.
Clinical effects:
Improved blood glucose control
Reduced appetite and caloric intake
Decreased body weight through reduced energy consumption
Stabilization of metabolic parameters
Potential positive effects on lipid profile and blood pressure
Clinical trials demonstrate significant body weight reduction (up to approximately 11 kg greater compared with GLP-1–based therapies over 26 weeks at higher doses).
Pharmaceutical quality:
Synthetic peptide with a precisely defined molecular structure
Selective dual action on GIP and GLP-1 receptors
High active ingredient purity
Stable injectable formulation
Manufactured in compliance with GMP standards
Dosage form:
Lyophilized white or off-white powder for preparation of a solution for injection.
After reconstitution, a clear, colorless solution free of visible particles is obtained.
Packaging:
1 vial × 10 mg
2 vials × 10 mg
Storage conditions:
After reconstitution, store refrigerated at 2–8°C (36–46°F) and use within 30 days.
Do not freeze.
Administration:
Reconstitution: Add 1.0 mL of bacteriostatic water → final concentration 10 mg/mL.
Weekly range: 2.5–15 mg once weekly (gradual dose escalation over 4-week intervals).
Measurement reference: At a concentration of 10 mg/mL, 1 unit (0.01 mL) = 0.1 mg (100 mcg) using a U-100 insulin syringe.
Dosage & Titration:
Recommended gradual weekly dose-escalation approach:
Initiation: 2.5 mg once weekly for 4 weeks to establish baseline tolerance.
Escalation: Increase by 2.5 mg every 4 weeks as tolerated.
Maintenance: 5–15 mg weekly depending on clinical response and tolerability.
Frequency: Once weekly (subcutaneous injection).
Timing: Administer on the same day each week at any time of day; rotate injection sites to minimize local irritation.
Laboratory Verification:
Certificate of Analysis (CoA):
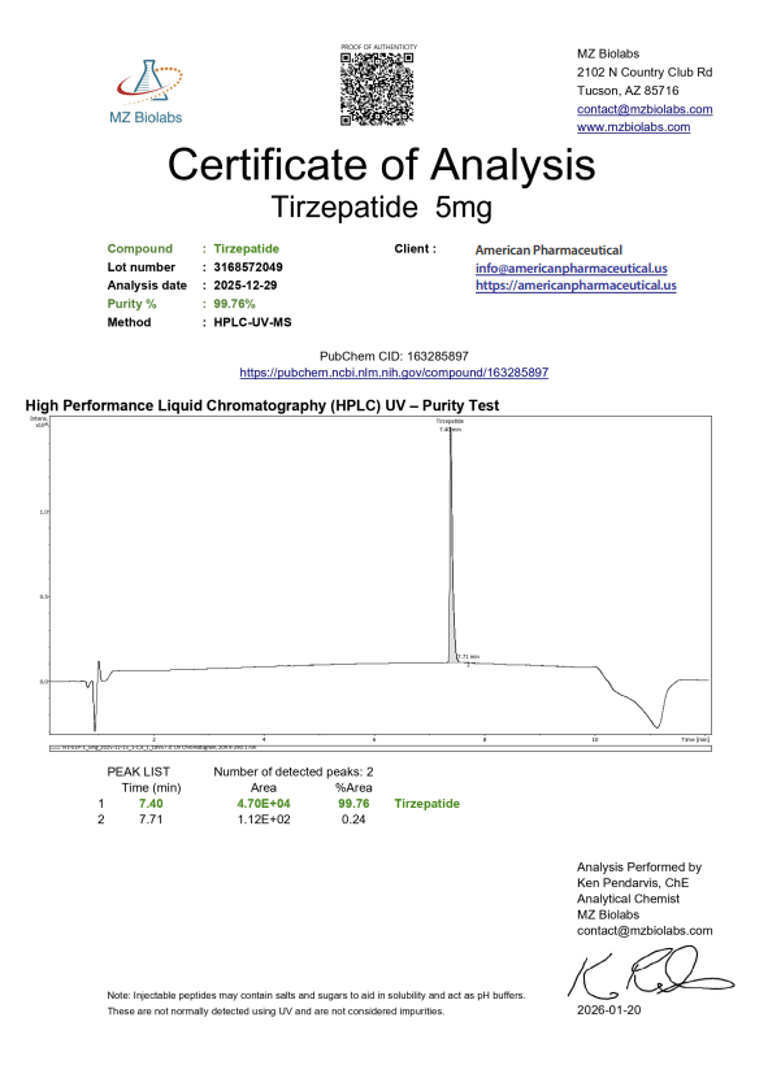 | 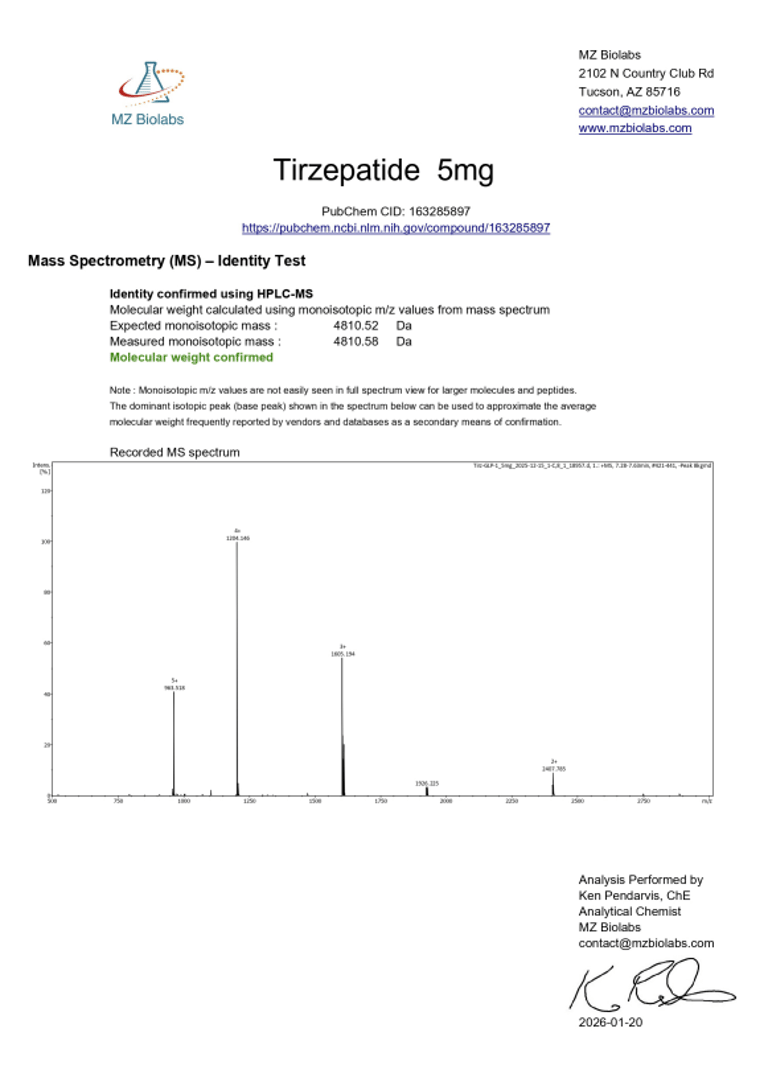 |
BioRegen:
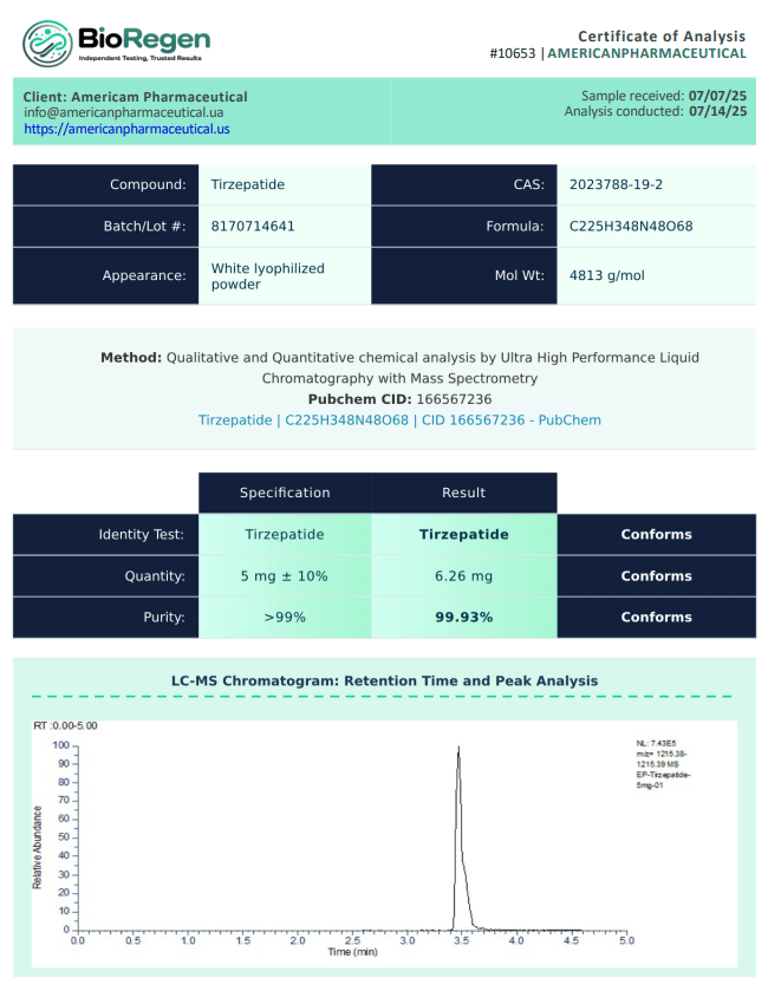 | 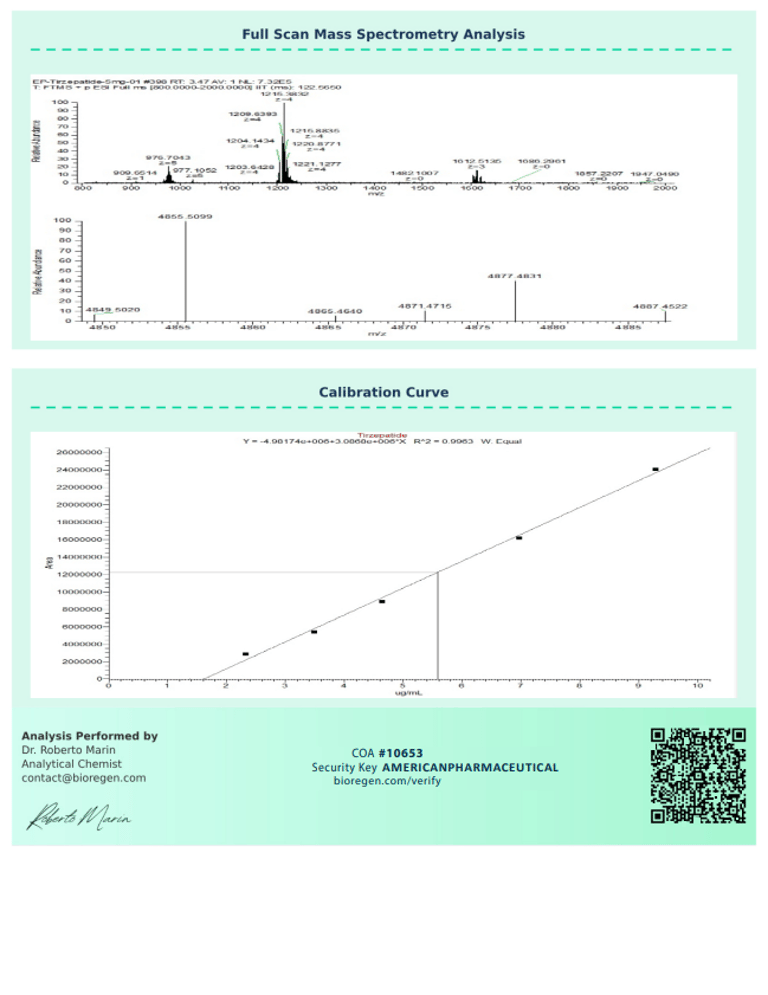 |
|---|
Manufacturer:
American Pharmaceutical, Inc.
Ohio, USA






