
SOMATROP
33.3 mg
Growth Hormone for Recovery & Physical Performance
SOMATROP is a pharmaceutical-grade recombinant human growth hormone developed in the United States for professional medical and scientific use. It is selected by athletes and active individuals for whom recovery, endurance, and physical performance quality are critically important.
When SOMATROP is selected:
During periods of intensive training and increased physical load
Throughout recovery following injury, surgery, or physical exhaustion
In cases of reduced regenerative capacity
To support lean muscle mass and overall physical condition
Potential effects under medical supervision:
Support of tissue and muscle recovery processes
Involvement in protein and lipid metabolism regulation
Contribution to the preservation of lean muscle mass
Participation in the development of physical endurance
Influence on recovery quality following physical exertion
Pharmaceutical quality:
Recombinant somatropin identical to endogenous human growth hormone
High degree of purification
Stable lyophilized formulation
Manufactured in compliance with GMP and FDA standards
Dosage form:
Lyophilized white or off-white powder for preparation of a solution for injection.
After reconstitution, a clear, colorless solution free of visible particles is obtained.
Packaging: 10 vials × 3.33 mg (10 IU each) — total 100 IU.
Storage conditions:
After reconstitution, store refrigerated at 2–8°C (36–46°F) and use within 30 days.
Do not freeze.
Administration:
Reconstitution: Add 1.0 mL of bacteriostatic water → final concentration 3.3 mg/mL (3300 mcg/mL).
Daily range: 150–500 mcg (conservative replacement protocols), up to 1000–2000 mcg (extended metabolic research protocols).
Measurement reference: At 3.3 mg/mL, 1 unit (0.01 mL) ≈ 33 mcg using a U-100 insulin syringe.
Dosage & Titration:
Recommended gradual weekly dose-adjustment approach:
Initiation: 660 mcg daily.
Conservative target dose: 660–990 mcg daily during the first 2–6 weeks.
Extended target dose: 1320–1650 mcg daily during weeks 7–13+ (metabolic/performance research protocol).
Frequency: Once daily (subcutaneous injection).
Course duration: Typically 12–24 weeks; may extend up to 48 weeks under appropriate medical monitoring.
Timing: Preferably administered in the evening or before bedtime; injection sites should be rotated systematically.
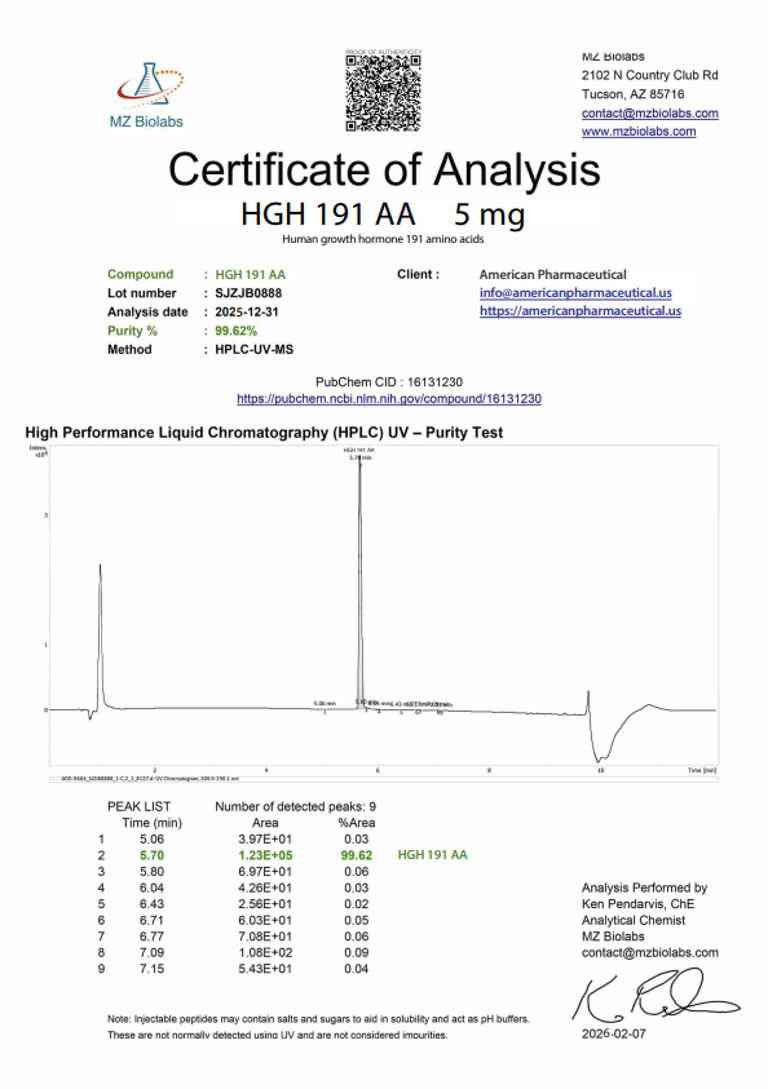
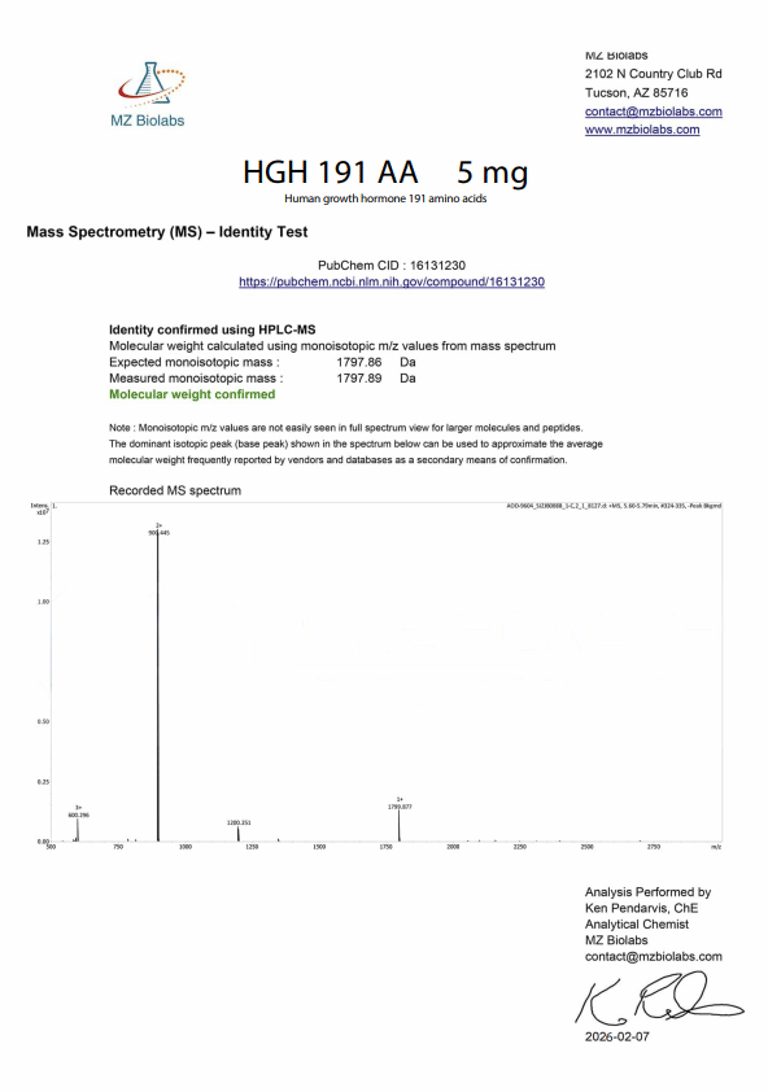
Laboratory Verification:
Certificate of Analysis (CoA):
 |  |
Manufacturer:
American Pharmaceutical, Inc.
Ohio, USA






