
SEMAGLUT
5 mg | 10 mg
Glycemic Control & Weight Management
SEMAGLUT is a recombinant DNA–engineered analog of human glucagon-like peptide-1 (GLP-1). It is used in medical practice for the treatment of type 2 diabetes mellitus and is recognized for its effects on blood glucose regulation, appetite control, and body weight management.
When SEMAGLUT is used:
In type 2 diabetes mellitus as an adjunct to diet and exercise
To improve glycemic control
Within treatment protocols aimed at reducing body weight and adipose tissue
In patients with elevated cardiovascular risk (within approved indications)
Pharmacological properties:
Semaglutide is a GLP-1 receptor agonist with high homology to the endogenous human hormone. Its action includes:
Glucose-dependent stimulation of insulin secretion
Suppression of glucagon secretion in the presence of elevated glucose levels
Delayed gastric emptying
Reduction in appetite and caloric intake
The use of semaglutide is associated with weight reduction and improvements in lipid profile parameters.
Clinical effects:
Reduction of fasting and postprandial glucose levels
Improved glycemic control (HbA1c)
Decrease in body weight and fat mass
Reduced appetite and cravings for high-calorie foods
Positive cardiovascular outcomes in patients with type 2 diabetes
Clinical studies have demonstrated significant body weight reduction (on average 10–15% of baseline body weight) in individuals with obesity.
Pharmaceutical quality:
Recombinant analog of human GLP-1
High biological purity
Stable pharmaceutical formulation
Manufactured in compliance with GMP standards
Dosage form:
Lyophilized white or off-white powder for solution for injection.
After reconstitution, a clear, colorless solution free of visible particles is obtained.
Packaging:
1 vial × 5 mg
2 vials × 5 mg
Storage conditions:
After reconstitution, store refrigerated at 2–8°C (36–46°F) and use within 56 days.
Do not freeze.
Administration:
Reconstitution: Add 2.0 mL of bacteriostatic water → final concentration approximately 2.5 mg/mL.
Weekly dose range: 250–2400 mcg (0.25–2.4 mg) once weekly (dose-escalation protocol).
Measurement convenience: At a concentration of 2.5 mg/mL, 1 unit (0.01 mL) ≈ 25 mcg using a U-100 insulin syringe.
Dosing & Titration:
Recommended dosing approach based on clinical studies and approved clinical data:
Weeks 1–4: 250 mcg (0.25 mg) once weekly to establish baseline tolerance
Weeks 5–8: 500 mcg (0.5 mg) once weekly
Weeks 9–12: 1000 mcg (1.0 mg) once weekly
Weeks 13–16: 1700 mcg (1.7 mg) once weekly
Weeks 17+: 2400 mcg (2.4 mg) once weekly as the target maintenance dose
The 2.4 mg once-weekly maintenance dose is FDA-approved for chronic weight management and represents the standard clinical approach.
Frequency: Once weekly (subcutaneous injection).
Timing: Administer on the same day each week at any time of day; rotate injection sites to minimize local irritation.
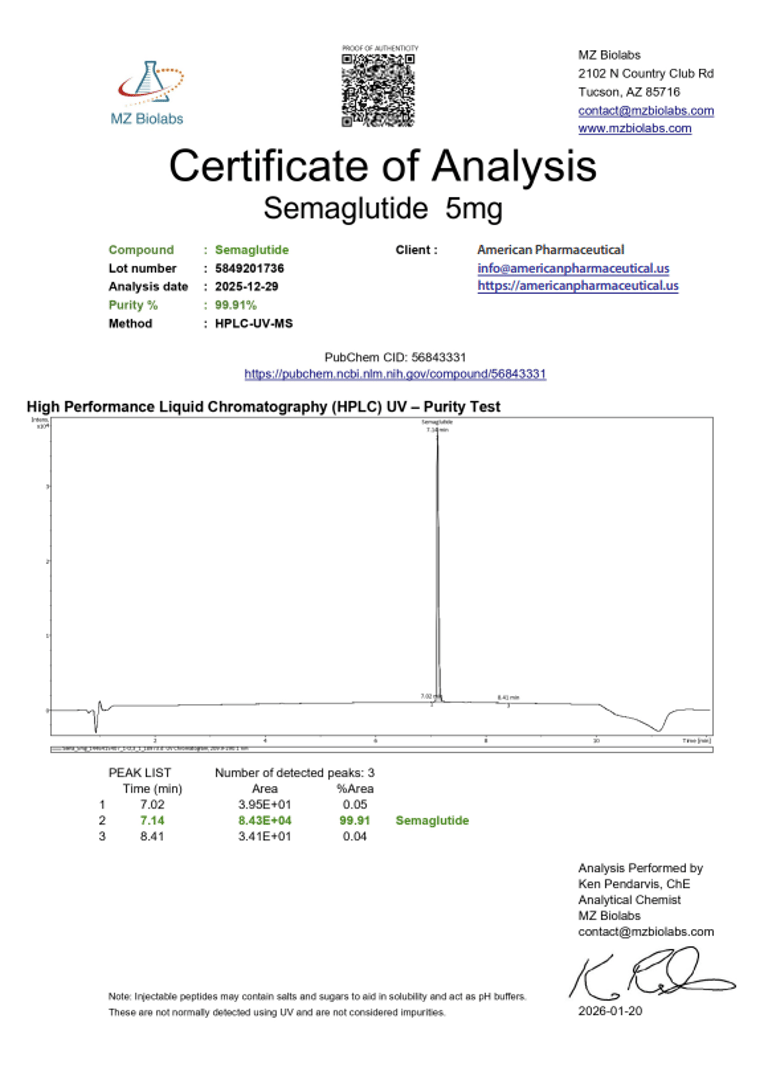
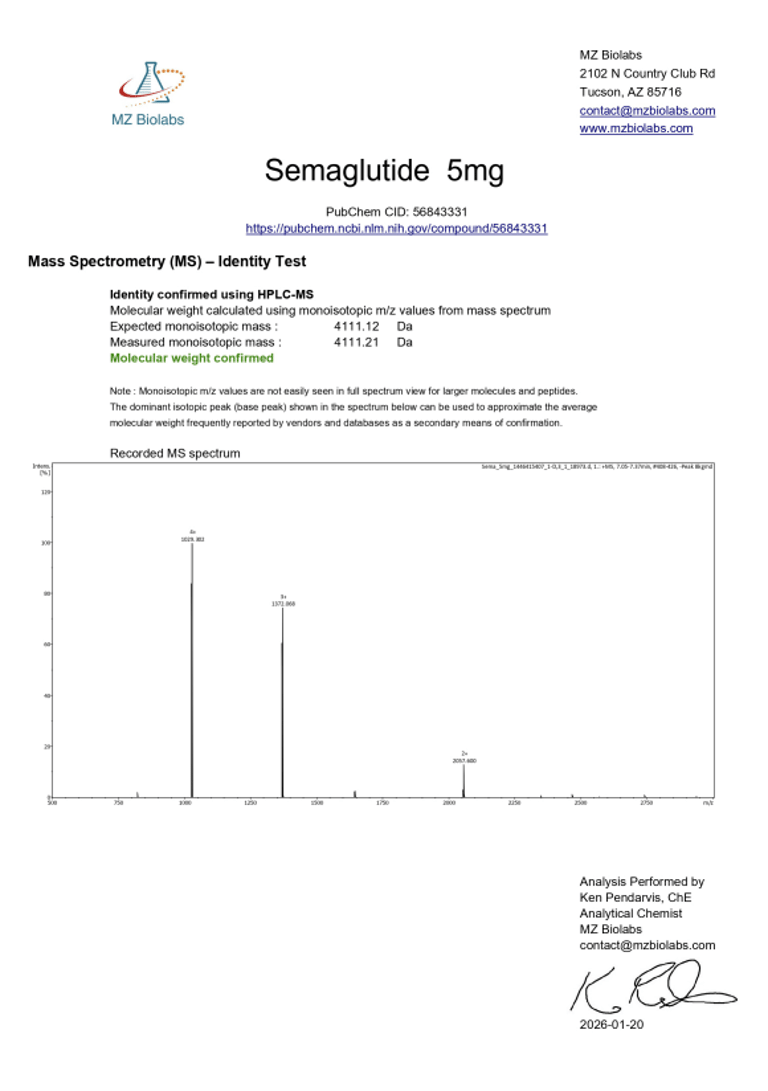
Laboratory Verification:
Certificate of Analysis (CoA):
 |  |
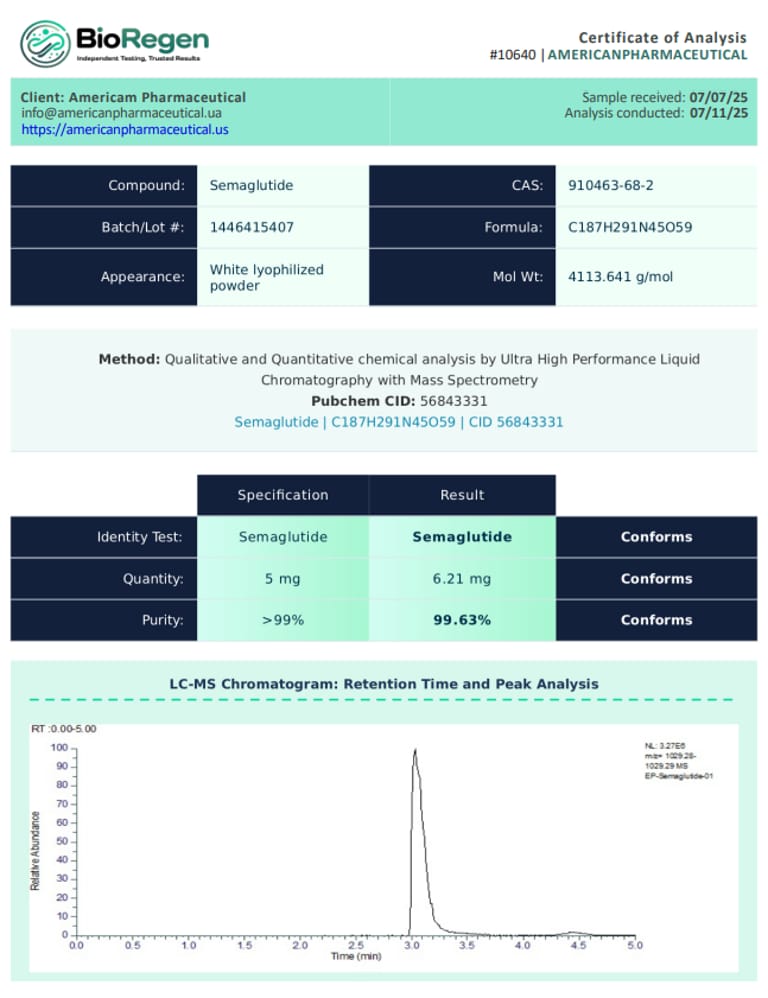
BioRegen:
 |  |
Manufacturer:
American Pharmaceutical, Inc.
Ohio, USA






