
RETATRUT
10 mg | 20 mg
Recovery, Metabolic & Body Composition Support
RETATRUT is a next-generation injectable peptide developed to support recovery, metabolic function, and body composition during periods of increased physical or metabolic demand.
When RETATRUT is selected:
Following injury or intensive training
In cases of overweight or obesity
During structured fat-reduction phases
In insulin resistance and carbohydrate metabolism disorders
For appetite regulation and eating behavior management
In metabolic syndrome or reduced insulin sensitivity
Key properties:
Supports recovery of muscle and connective tissues
Participates in the regulation of metabolic processes
Delays gastric emptying
Reduces appetite and caloric intake
Promotes recovery following physical exertion
Clinical effects:
Reduction of body weight primarily through fat mass loss
Improved glycemic control
Increased insulin sensitivity
Decreased appetite and food cravings
Positive influence on overall energy balance
In clinical studies, participants receiving retatrutide at 12 mg weekly achieved an average body weight reduction of approximately 24% over 48 weeks. In adults with type 2 diabetes, retatrutide (up to 12 mg weekly) resulted in approximately 17% body weight reduction over 36 weeks.
Pharmaceutical quality:
Synthetic peptide with a precisely defined molecular structure
High degree of purification
Stable pharmaceutical formulation
Manufactured in compliance with GMP standards
Dosage form:
Lyophilized white or off-white powder for preparation of a solution for injection.
After reconstitution, a clear, colorless solution free of visible particles is obtained.
Packaging:
1 vial × 10 mg
2 vials × 10 mg
Storage conditions:
After reconstitution, store refrigerated at 2–8°C (36–46°F) and use within 38 days.
Do not freeze.
Administration:
Reconstitution: Add 1.0 mL of bacteriostatic water → final concentration 1.0 mg/mL.
Weekly range: 2–12 mg once weekly (gradual escalation over 12+ weeks).
Measurement reference: At 10 mg/mL, 1 unit (0.01 mL) = 0.1 mg (100 mcg) using a U-100 insulin syringe.
Dosage & Titration:
Recommended gradual weekly dose-escalation approach:
Initiation: 2 mg once weekly during the first 4 weeks to establish baseline tolerance.
Escalation: Increase to 4 mg weekly (weeks 5–8), then to 8 mg weekly (weeks 9–12).
Maintenance: 12 mg weekly (from week 13 onward) for maximal efficacy; 8 mg weekly may serve as an alternative maintenance dose.
Frequency: Once weekly (subcutaneous injection).
Cycle duration: Minimum 12 weeks. Clinical studies have been conducted for up to 48 weeks, demonstrating sustained weight reduction.
Timing: Administer on the same day each week at any time; rotate injection sites to minimize local irritation.
Laboratory Verification:
Certificate of Analysis (CoA):
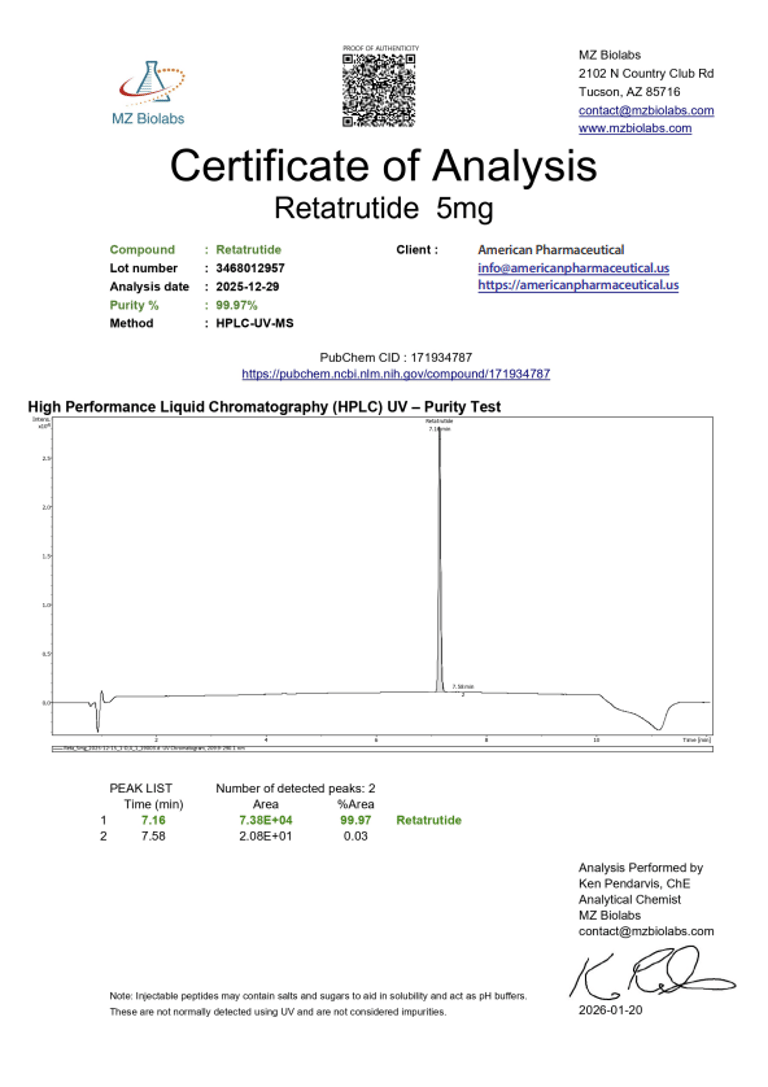 | 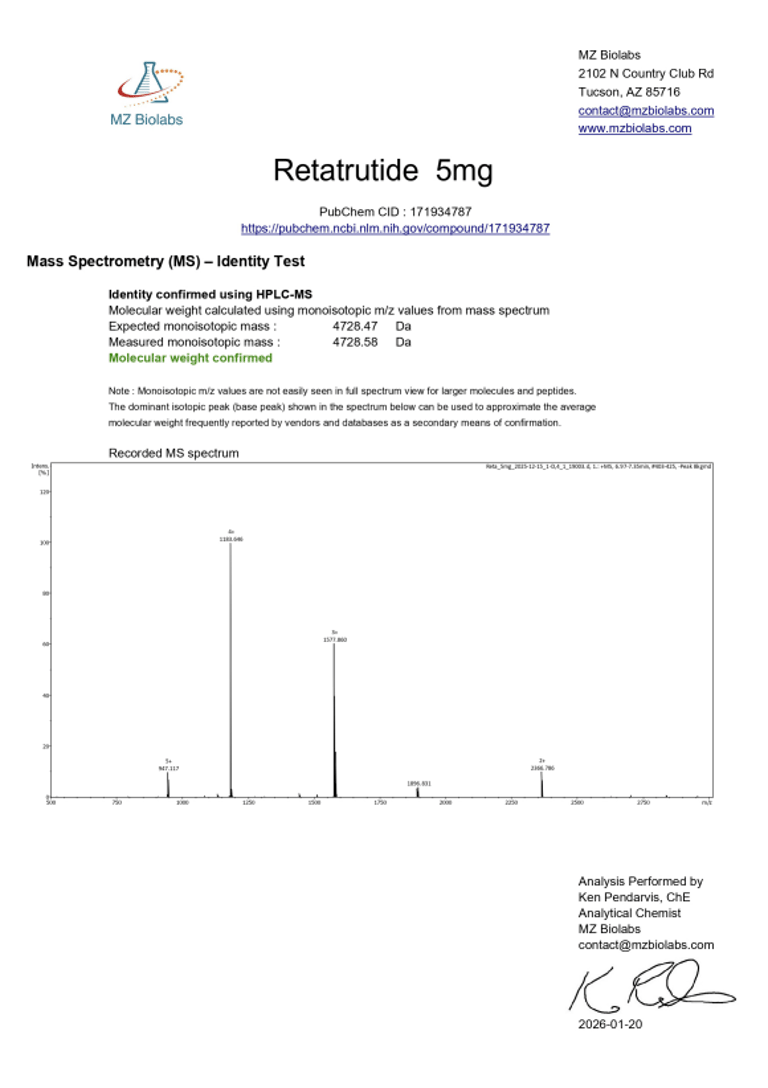 |
BioRegen:
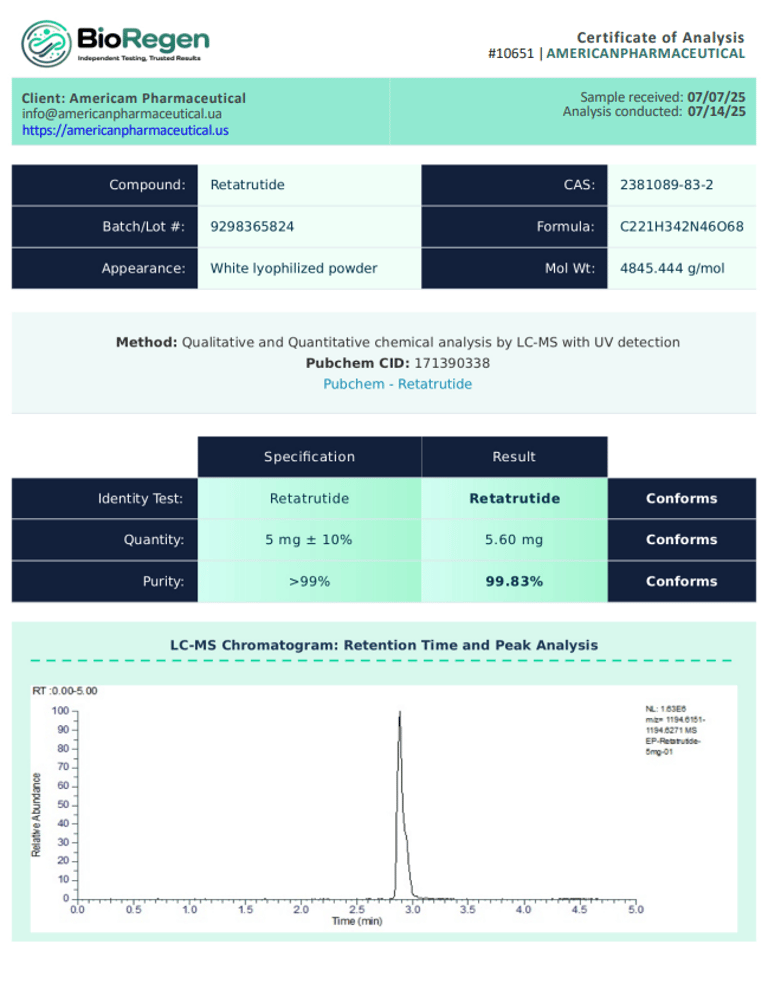 | 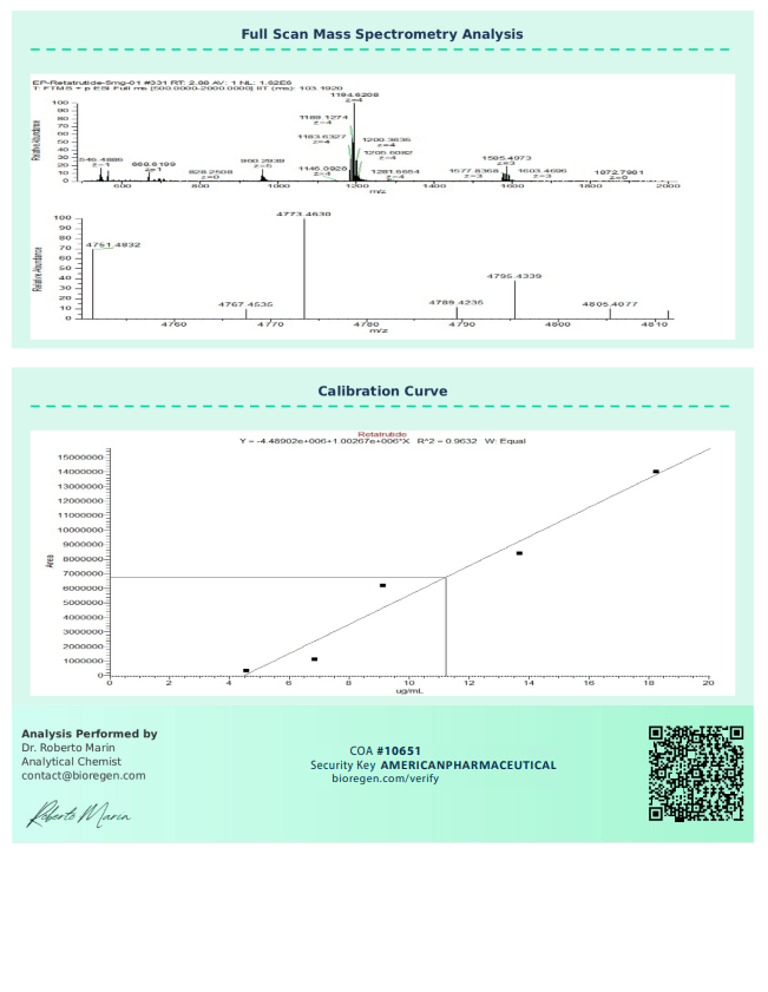 |
Manufacturer:
American Pharmaceutical, Inc.
Ohio, USA






